The at-home kits were divvied up and picked up by 19 different school districts in Erie County for distribution.

ABOUT US
A TRUSTED PARTNER IN
GLOBAL HEALTH
Access Bio’s mission is to improve the lives and well-being of people around the world. Through the development of in vitro diagnostics technology, Access Bio has successfully commercialized the highest quality products to battle malaria and other serious diseases.
We are a trusted partner to international public health agencies and organizations, including the World Health Organization, the Bill and Melinda Gates Foundation, UNICEF, and many others.
PRODUCTS

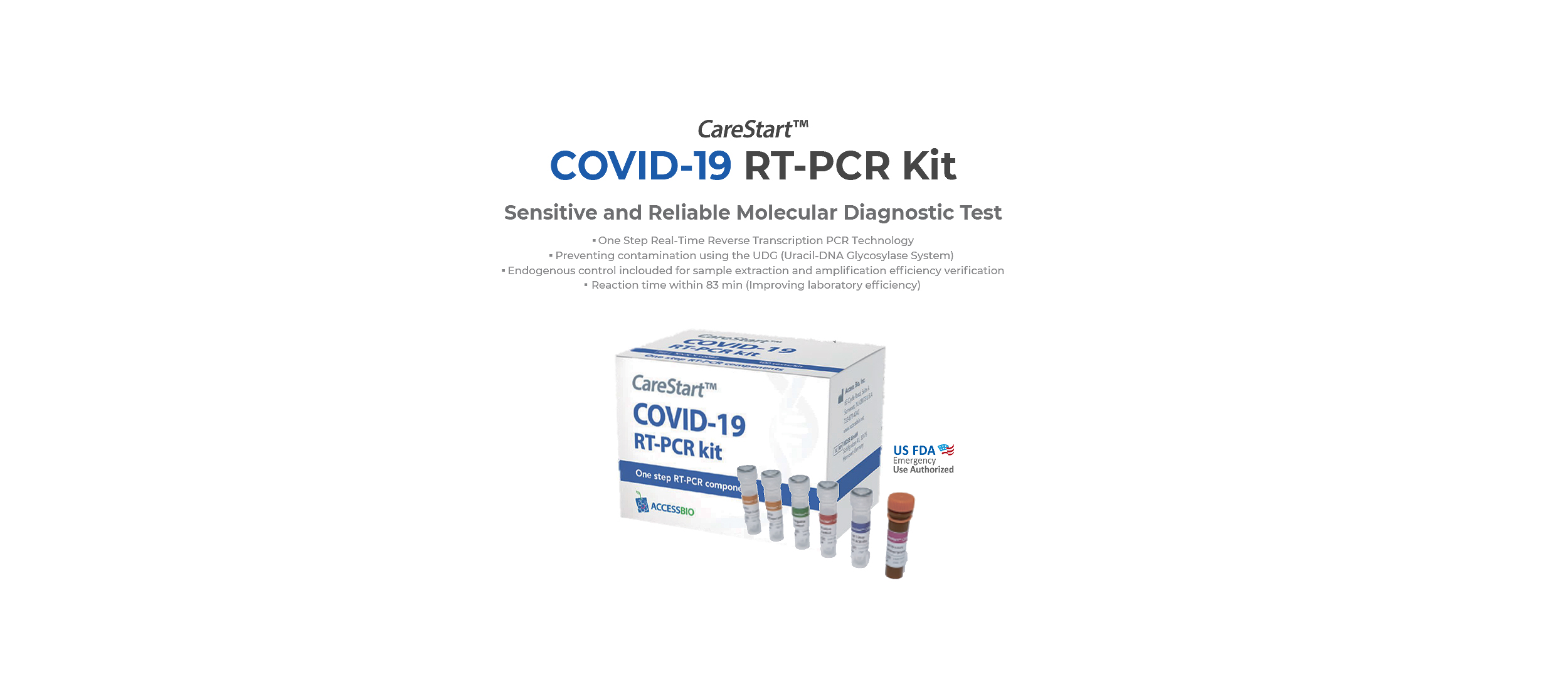
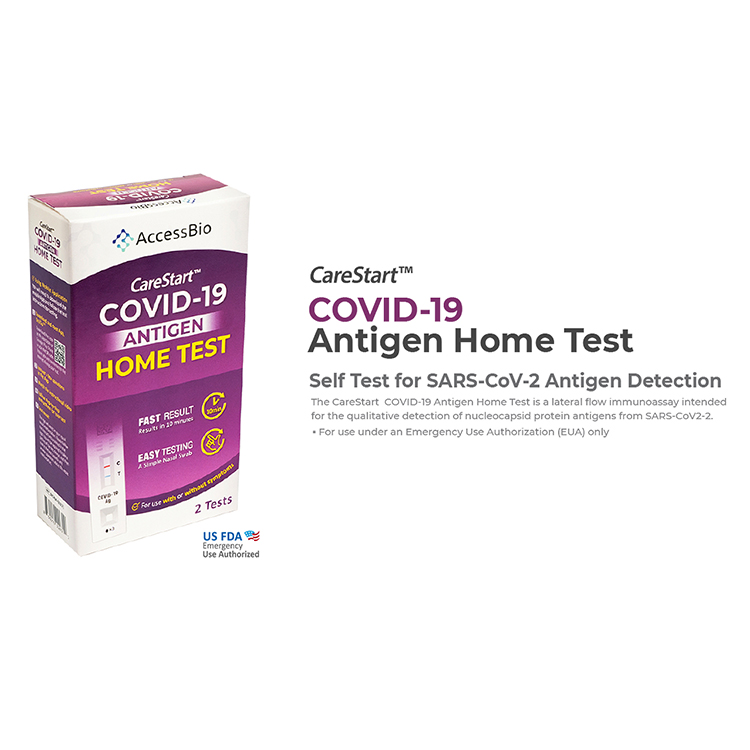
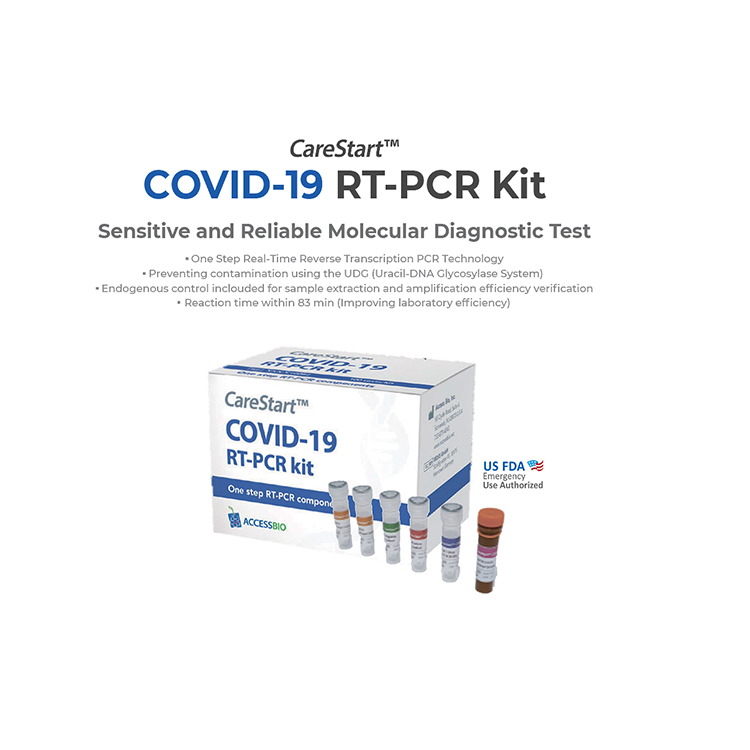
COVID-19
Diagnostic
Tests

Rapid
Diagnostic
Tests

Analyzers

Molecular
Tests
THE RANGE
LEADING THE FIGHT
AGAINST COVID-19
A challenge as complex as the Covid-19 pandemic requires
innovative, adaptable solutions. To meet the need for accurate,
reliable screening, we have a suite of products that includes
PCR, serology, and antigen testing.
NEWS
GLOBAL
OPERATIONS
AND REACH
ACCESSBIO
INVESTORS
WE ARE
HIRING
GET IN TOUCH
CONNECT WITH
US HERE AND
WE’LL BE TOUCH
We are always seeking to collaborate with forward-thinking individuals, organizations, and institutions.